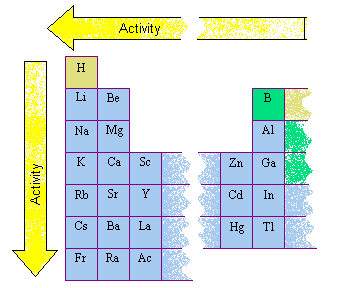
Germanium lies within the diagonal line marking the boundary between metals and nonmetals, so it should be a metalloid.Magnesium lies to the left of the diagonal line marking the boundary between metals and nonmetals, so it should be a metal.Explore the periodic table of the chemical elements using. Interactive periodic table with all properties. Know all element Facts, Electronic Configuration, oxidation state, isotopes. Upto Date latest full Modern Periodic Table with dynamic layouts. The Arabic numbering system is the most widely accepted today.\), selenium lies above and to the right of the diagonal line marking the boundary between metals and nonmetals, so it should be a nonmetal. Dynamic Interactive Periodic Table of Elements with names, symbols, atomic mass - Chemical, Physical, Atomic and Thermal Properties & Trends. Many periodic tables include both Roman and Arabic numbers. The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.The CAS system used letters to differentiate main group (A) and transition (B) elements.The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.Then, they must color-code the alien table using the family names. They must use the extraterrestrial data to arrange the alien elements onto the blank periodic table. Three systems have been used to number families and groups: Students are given some background information, a list of clues, a blank periodic table, and a ‘word bank’ of the alien periodic table squares. Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. Noble Gases: - Group 18 (VIIIA) - 8 valence electrons.The periodic table has a total of 118 elements. It exemplifies humanity’s attempt to decipher the underlying building components of our reality. Halogens: - Group 17 (VIIA) - 7 valence electrons The periodic table reveals the exquisite tapestry of matter’s diversity, from the noble gases’ passive nobility to the alkali metals’ unexpected reactivity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
