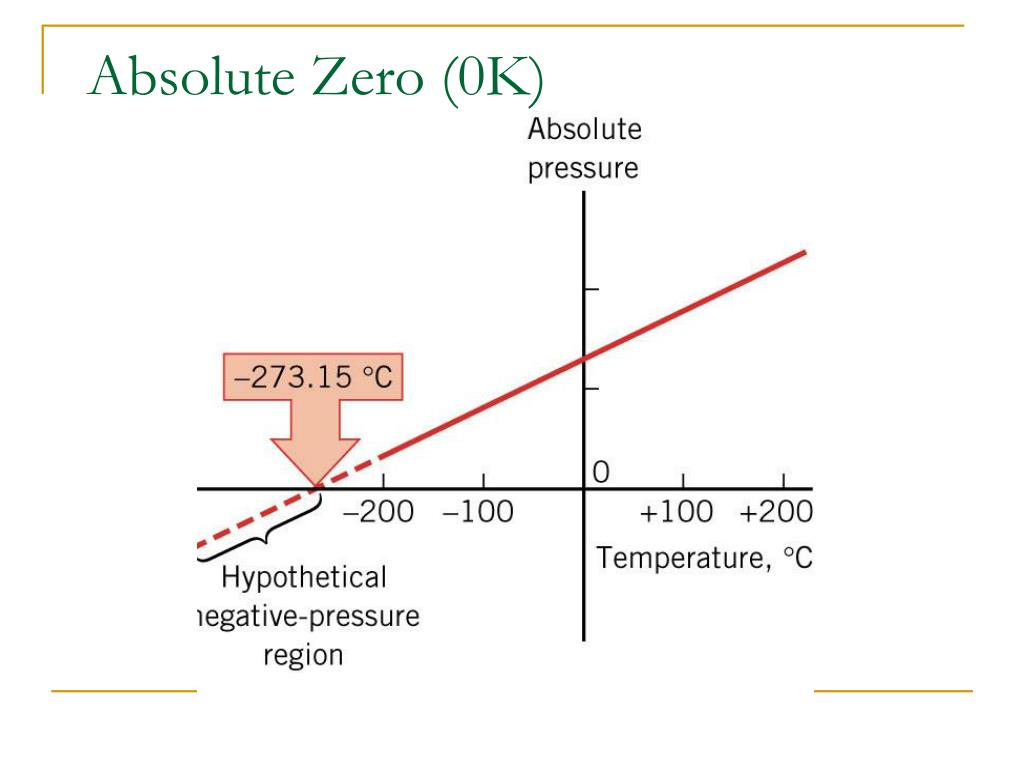
I hope that answers some of your questions on Absolute Zero. Absolute zero, 0 Kelvin, -459.67 Fahrenheit, or -273.15 Celsius, is the minimum possible temperature: the state in which all motion of the particles in a. The electrons for example will always have some kinetic energy, even if it is simply a small vibration. So, from these and other tests, physicists believe that even if we get close to absolute zero, it is never actually attainable because the atoms of an element must always be in some type of motion. As the gas is cooled, it will make a phase transition from gas into. At this temperature, the gases these scientists were experimenting on became interlaced in such a way that they were no longer gas, liquid, or solid, they were something else, never before seen. First of all, the gas will no longer be a gas at absolute zero, but rather a solid. The coldest observable temperatures that have been reached are within a fraction of a degree of 0 Kelvin. 
To have something at absolute zero in theory means it has no kinetic energy, but that’s not possible, because then the materials would cease to exist as normal matter, due to laws of quantum mechanics. Nernst proposed that the entropy of a system at absolute zero would be a well-defined constant. 
Now, just because we can say absolute zero exists, does not mean we actually can reach or observe it. In Kelvin, which is what most physicists like to use, 0 is absolute zero. There are three fairly popular measurements for gauging temperature. These all factor into the temperature of stuff. 
Molecules have different ways of moving around, they can vibrate, the atoms rotate, and they can have a collective motion, similar to a wave of water. The Third Law defines the behaviour of entropy at absolute zero. For example, if the molecules in this room were moving around very slowly, it would be very chilly in here. Temperature is a gauge for how much energy those molecules have, and the more they have, the hotter we say something is. In fact, they are always randomly moving around, and we measure that with temperature. Absolute zero is used in some perpetual motion devices.Hi and welcome to this video lesson on absolute zero.īefore we jump into what absolute zero is and whether or not it can be reached, let’s first discuss the idea of temperature in general. At absolute zero temperature the thermal motion ceases and the position of the molecules are fixed/certain in pure crystalline substance, defining the absolute zero entropy (the Third Law of Thermodynamics). Once the quantum dimension is understood, technology achieving absolute zero is invented. The old laws of thermodynamics indicate that absolute zero cannot be reached. Cryonics is used by the occupants of world ships travelling at sub-light speed where long journeys would outlive them and the technology for faster-than-light has not yet been invented. superfluidity - where a fluid has zero viscosity and therefore flows without any loss of kinetic energy - and can even flow uphillĬryogenics is the production and behaviour of materials at very low temperatures, compared to cryonics which is the cryopreserving of humans and animals with the intention of future revival.superconductivity - where electrical resistance vanishes and magnetic flux fields are expelled from a material - used for example in powerful electromagnets and vactrains.Bose–Einstein condensate - the fifth state of matter.Entropy, the disorderliness or change in enthalpy of a system, is zero or even nothing.Īt temperatures close to absolute zero, matter exhibits quantum effects such as: The third law, in its modern formulation, asserts that the absolute entropy tends to zero, in the limit as temperature tends to absolute zero, for each aspect. Enthalpy, the total heat content of a particular system, is zero. Absolute zero is the lowest temperature possible at zero kelvins (−273.15° celcius), where the fundamental particles of nature have only zero-point energy particle motion via quantum fluctuations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
